Background
Historically, the use of support surfaces in the treatment of myocutaneous flaps resulting from Stage IV pressure ulcers on the torso has been limited in our hospital to air fluidized therapy. The WOC nurses were asked to find an alternative surface for post-myocutaneous patients. The professional staff recognized multifactorial concerns with the use of the fluidized therapy beds. These concerns included staff injuries with patient transfers and patient safety.
Methods and Results
In our search to find a different support system, we initially trialed a mattress that provided immersion therapy. Immersion therapy simulates a fluid environment and immerses the patient into the surface. During this evaluation, patients experienced adverse outcomes due to the microclimate of the patient’s skin. Patients had problems with moisture and skin breakdown.
In an attempt to manage the microclimate environment in these post-myocutaneous patients, we trialed a new prototype Immersion therapy mattress with low air loss. Our five case studies consisted of post-myocutaneous flap patients from stage IV torso ulcers. The study was from Sept. 2013 to Feb. 2014. All five patients were placed on the low air loss Immersion therapy mattress directly after surgery along with our hospital’s total pressure ulcer management plan. As part of our hospital’s clinical pathway for post-myocutaneous flap patients, all five patients were transferred to a critical access hospital. The low air loss Immersion therapy mattress was transferred with the patient. Upon discharge to home, all flap incisions were approximated and closed. Hospitalization days between both hospitals ranged from 4 – 7 ½ weeks.
Patient 1
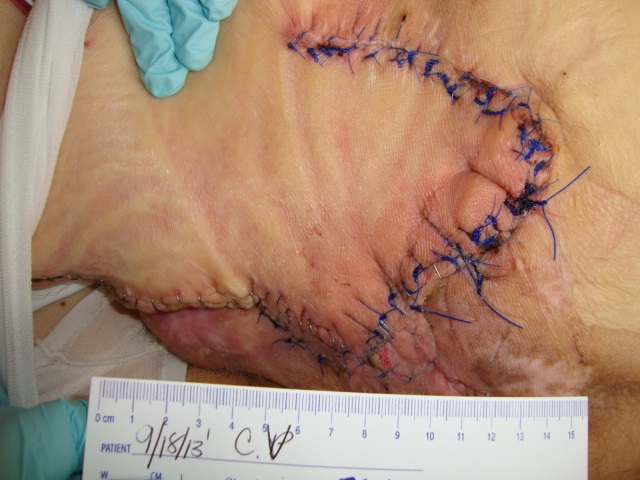
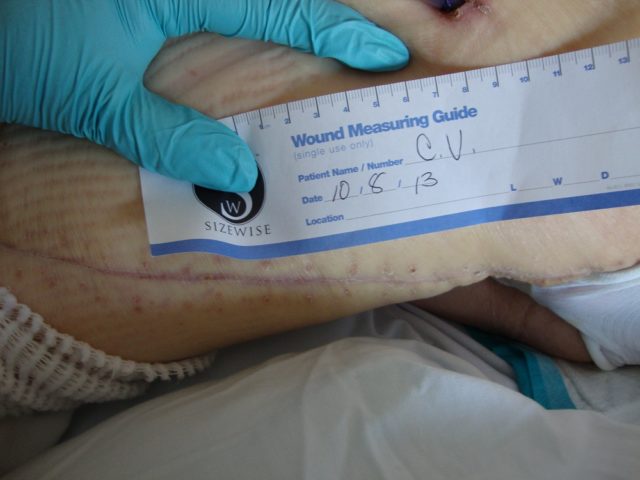
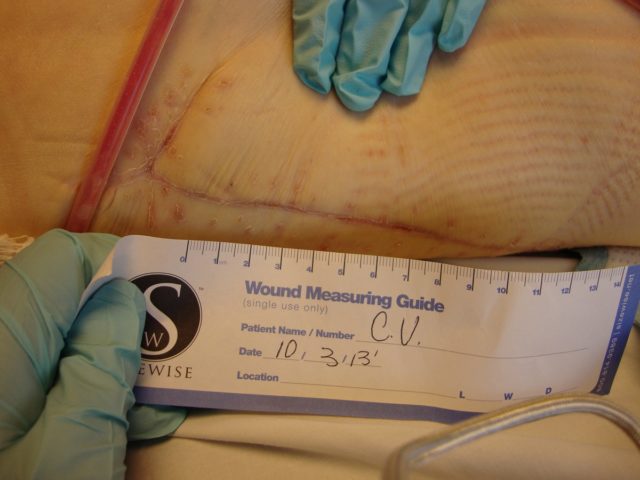
Patient 2
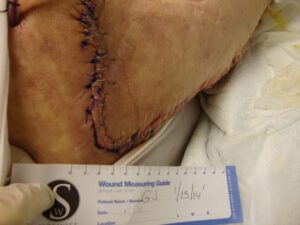
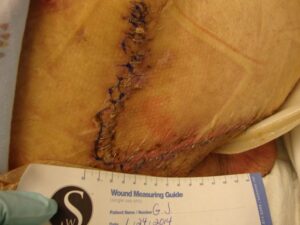
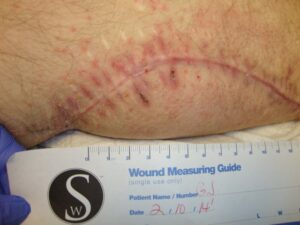
Patient 3
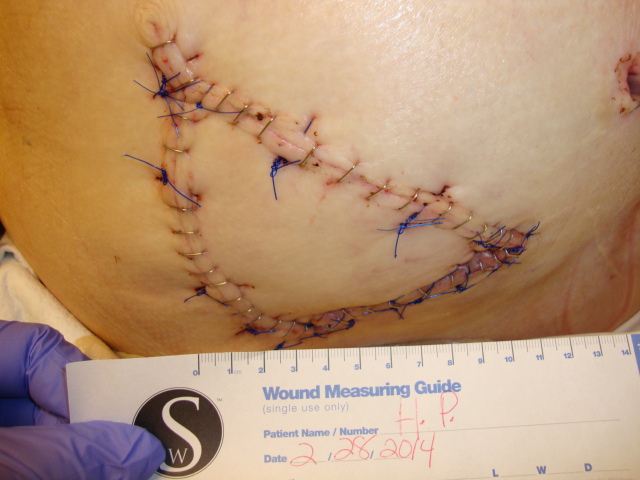
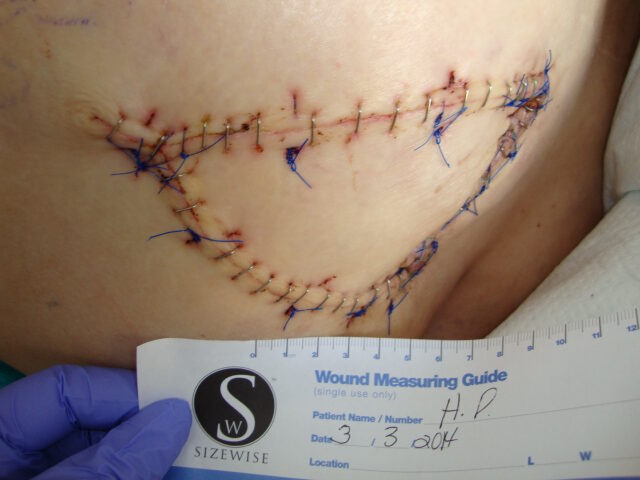
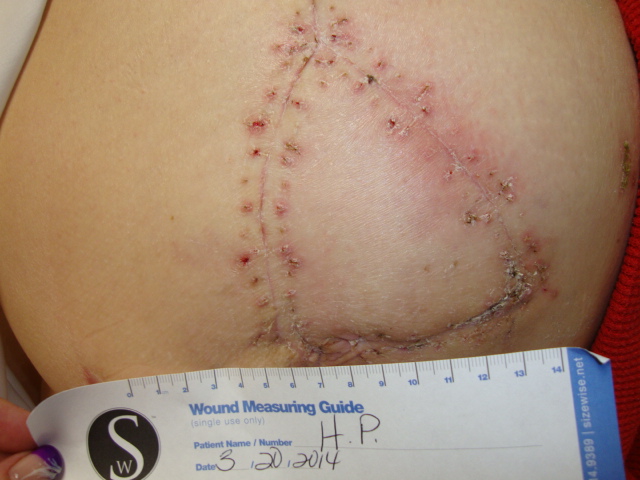
Patient 4
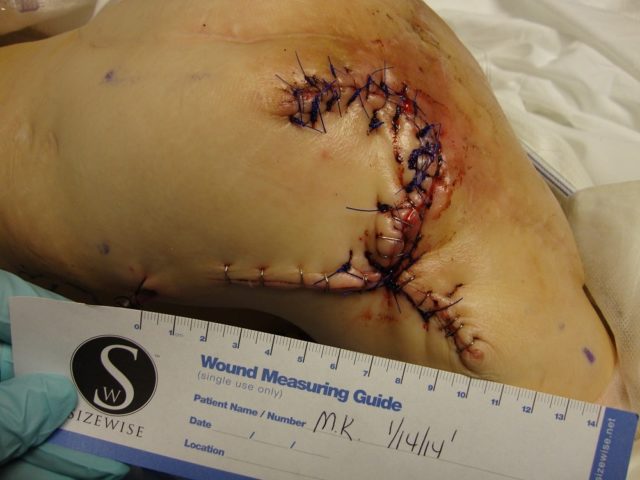
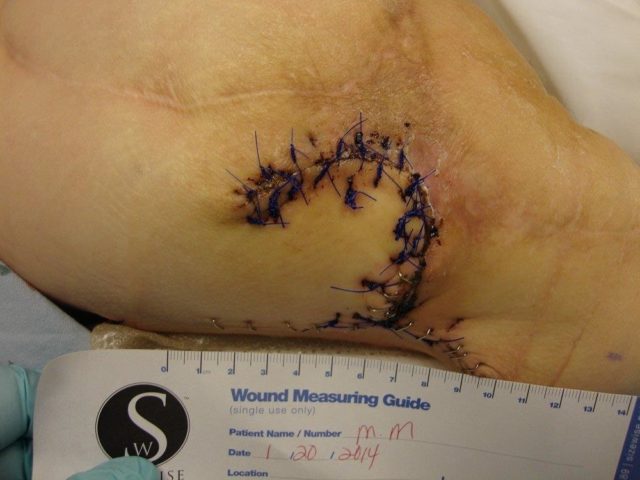
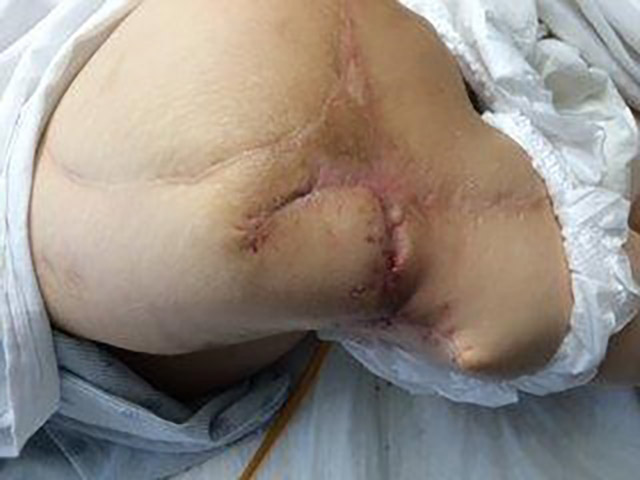
Patient 5
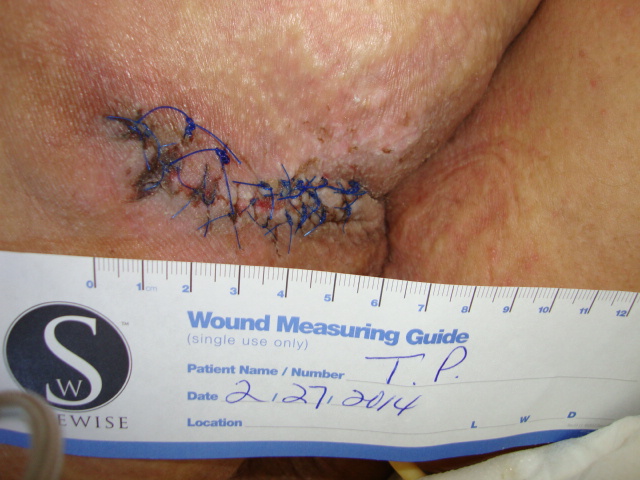
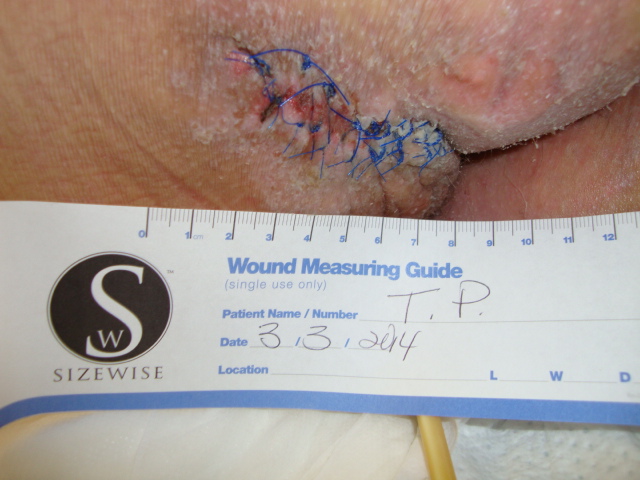
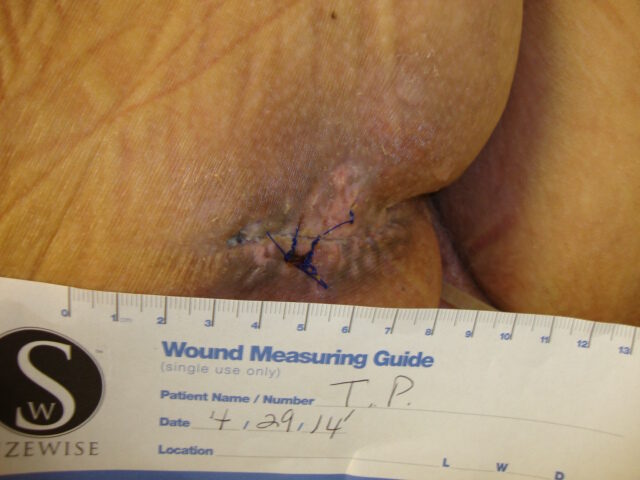
Conclusion
Our experience with this newer therapy was quite positive for patient outcomes; and feedback from our patients was favorable. As a result of this study, our hospital no longer uses air fluidized therapy for post-myocutaneous flaps. The response from our multidisciplinary team has been supportive of this change.

